Overcoming Clinical Trial Recruitment Obstacles With Digital Advertising

As patients become increasingly empowered, sponsors and CROs need to update their recruitment tactics accordingly.
Right now, the clinical trial industry is contending with a serious problem: the disconnect between the number of Americans who have an interest in participating in a clinical study, and the number who actually end up doing so.
Part of the issue is can be attributed to a general lack of awareness. For example, according to the NIH, approximately 85% of cancer patients remain unaware of active clinical studies, yet 75% would be willing to participate if they knew they existed.
Another recent poll conducted by Zogby Analytics and Research!America suggests that 72% of patient respondents would likely enroll in a trial based on their doctor’s recommendation; however, only about 22% indicated that they’d ever been spoken to about clinical trial opportunities by a medical professional.
Interestingly, recently published Tufts CSDD research found that 77% of nurses and 90% of physicians consider themselves “somewhat” or “very” comfortable discussing clinical trials with their patients, and yet less than 0.2% of these providers refer patients to active trials.
According to the same report, the root of this problem can be traced to a lack of communication and transparency with primary health care providers — an issue that needs to be addressed by the entire clinical research community. That said, the rise of search engines, social media, and the empowered patient have presented sponsors and CROs with an unprecedented opportunity to reach their trial’s target patient base directly. This in turn has placed them squarely in the driver’s seat when it comes to improving recruitment efficiency and keeping enrollment timelines on track.
Tap Into the Largest Patient Database In Existence
When it comes to digital advertising, Google’s user base still reigns supreme. Last year, the search engine giant processed more than 28 billion searches on healthcare-related topics in the United States alone, 28% of which were resolved by medical ads. In total, search ads amassed a whopping one billion clicks. This represents a massive opportunity for clinical trials looking to capture the attention of “active” patients, or those patients who have taken control of their decision-making process and are actively looking for treatment options, which makes them more likely to convert.
Leverage Social Media’s Massive Audience
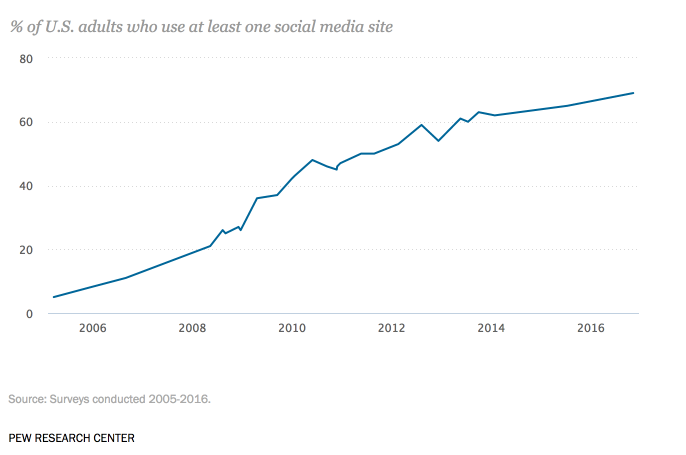
As social media usage continues to soar, marketers across nearly every industry have begun to recognize its potential as an impactful advertising channel. Today, approximately 69% of U.S. adults are active on at least one social media site, representing a 64% spike over the past decade.
In particular, Facebook’s immense audience and powerful targeting capabilities make it ideally suited for clinical trial recruitment. Tap into the platform’s 1.86 billion base of active monthly users and target based on highly specific demographic criteria, including age, gender, location, interests, behavior, and more. You can also leverage Facebook’s Custom Audiences, Lookalike targeting capabilities, and the Facebook Audience Network to further expand the reach of your campaign.
Improve Your ROI Through Active Campaign Management
Both Facebook and Google AdWords provide a comprehensive set of data analytics and performance monitoring tools. Of course, all advertising materials will need to be submitted in advance for IRB approval; however, through proactive monitoring and campaign management (e.g., A/B testing, modifying targeting based on real-time data, scaling spend in accordance with demand, etc.), sponsors and CROs often see total cost savings to the tune of 64%.
At the end of the day, as patients become increasingly empowered by digital media, relying exclusively on the medical community and traditional advertising methods to generate qualified patient referrals is no longer an effective approach. Implementing a strategically sound and actively monitored digital recruitment strategy will ultimately generate more qualified candidates at a significantly reduced cost.

 Back to Blog Home
Back to Blog Home