What Kind of Impact Can Homecare Have on Clinical Trial Recruitment and Retention?

In an increasingly patient-centric clinical environment, sponsors are finding they can improve enrollment outcomes by bringing the trial directly to the patients.
As the impact of social media, mobile devices, and other medical technologies continues to swell, a new patient-centric clinical landscape is emerging. In other words, more and more Pharma companies are actively soliciting input from trial subjects, then incorporating those insights into the trial’s design.
What exactly does this mean in practical terms? Sponsors need to seek out new technologies and methods to make trials fit more seamlessly into patients’ day-to-day lives, thereby improving recruitment, retention, adherence, and ultimately, their bottom line.
I recently came across an interesting article, co-authored by Helena Baker and Matimba Swana, about the economic impact of clinical homecare, or the practice of “bringing clinical research procedures to the subject, rather than the traditional method that dictates the subject goes to the investigator site.” Baker and Swana outline a wide variety of the potential benefits that homecare can bring, but of particular interest to me was the impact it can have on patient recruitment and retention outcomes. Here are a few of the key takeaways.
Homecare Improves Clinical Recruitment and Retention
“Site fatigue” is one of the biggest barriers to enrollment efficiency – investigator sites become overwhelmed, either by too many patients or simultaneous trials, which can have a detrimental effect on recruitment and retention results. Sending personnel directly to patients in their homes to administer care reduces the volume of patients visiting the site, which reduces the overall strain for on-site personnel and resources.
Baker and Swana point to a 2012 study of a longitudinal randomized controlled study in which “25% of the trial’s participants required home visits, which led to a 4% overall dropout rate.” Studies that involve geriatric patients with a “high medical burden” and those with a “large number of complications” run a much higher risk of losing patients due to issues surrounding on-site checkups.
For comparison’s sake, a sponsor conducted a two-year oncology study that required regular (bi-weekly) blood samples from 63 participants spread across six sites in the UK. Five out of six sites offered homecare as an option – patient recruitment rates increased by 63%, and retention hit more than 98% (one patient was forced to drop out after becoming pregnant).
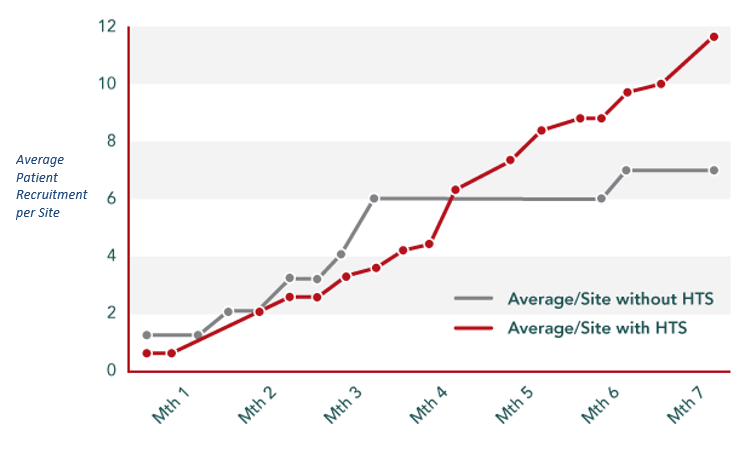
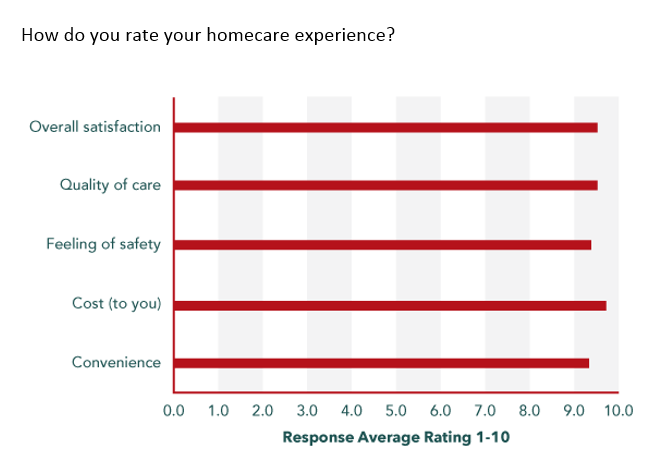
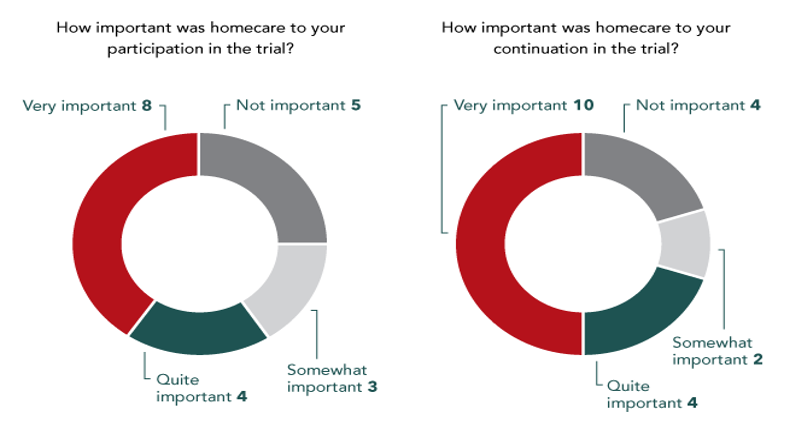
Similarly, another long-term oncology study that “required compounding within 24 hours of drug administration for 57 patients across three UK sites” used homecare and experienced similar results. The patient retention rate soared to 98%, with only one patient dropping out over the course of the study. The overwhelmingly positive results of the post-trial patient assessment are outlined in the charts below:
A Tech-Forward Approach to Homecare
In an increasingly patient-centric environment, homecare has clearly emerged as a win-win for participants and sponsors alike; however, as mobile and wearable technologies continue to proliferate, many in the industry are recognizing their potential to improve recruitment and retention outcomes across the board.
Major Pharma companies, such as Pfizer, Astrazeneca, and GlaxoSmithKline, are already investing in Mobile health, or “mHealth,” programs that extend across the entire R&D process. Similarly, sponsors are taking advantage of sensor technology to remotely monitor patients’ vitals, key environmental data, and even drug adherence.
At the end of the day, if sponsors want to see a real reduction in enrollment-related costs, they need to start designing their trials with the needs and/or limitations of their subjects in mind. It’s not just the smart thing to do – it’s the right thing as well.

 Back to Blog Home
Back to Blog Home